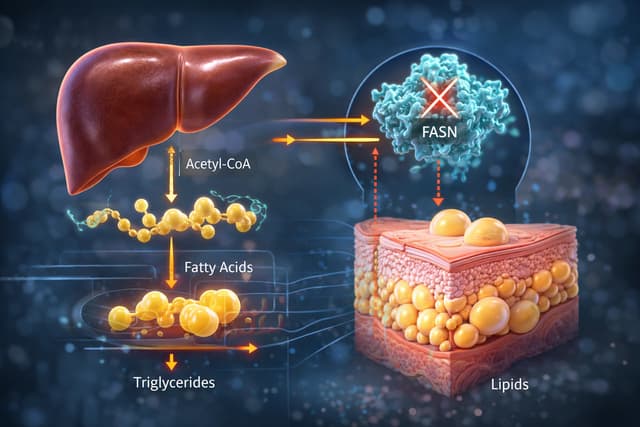
Sagimet Biosciences (NASDAQ:SGMT), a clinical-stage biopharmaceutical company developing novel fatty acid synthase (FASN) inhibitors, reported its fourth-quarter and full-year 2025 financial results on Wednesday.
The company highlighted a robust liquidity position and significant clinical momentum, specifically within its metabolic dysfunction-associated steatohepatitis (MASH) and dermatology franchises.
Sagimet ended the year with a cash balance of $113.1 million, providing a substantial runway to fund its upcoming clinical catalysts.
A major highlight of the year was the successful completion of a Phase 1 pharmacokinetic (PK) trial evaluating the co-administration of denifanstat, the company’s lead FASN inhibitor, with resmetirom.
The study reported no significant safety signals, clearing a critical path for potential combination therapies.
To further solidify this strategy, the company secured an exclusive global license to TAPI resmetirom API.
In the dermatology sector, Sagimet reached a major regulatory milestone in China.
The National Medical Products Administration (NMPA) accepted a New Drug Application (NDA) for denifanstat for the treatment of acne.
Building on this momentum, the company is preparing to launch Phase 2 studies for its second asset, TVB-3567, specifically targeting acne in 2026.
The company’s most anticipated milestone is a Phase 2 proof-of-concept trial in F4 MASH—the most severe stage of the disease before liver failure—which is slated to begin in the second half of 2026.
This trial aims to address the urgent medical need for patients with advanced fibrosis, a segment where few therapeutic options currently exist.